
Graphite - Definition, Structures, Applications, Properties, Use with Videos and FAQs of Graphite
Graphite - Graphite is an allotrope of carbon. It is a soft, slippery, greyish black substance. It has a metallic lustre and is opaque to light. Graphite is a good conductor of heat and electricity. To learn the Applications, Structures, Properties, Use with Videos and FAQs of Graphite, Visit BYJU’S for more information.
Graphite - Graphite is an allotrope of carbon. It is a soft, slippery, greyish black substance. It has a metallic lustre and is opaque to light. Graphite is a good conductor of heat and electricity. To learn the Applications, Structures, Properties, Use with Videos and FAQs of Graphite, Visit BYJU’S for more information.

Draw the structure of graphite and diamond. Explain how the structures are responsible for the difference in their physical properties.
Is carbon and graphite the same thing? - Quora
Is carbon and graphite the same thing? - Quora
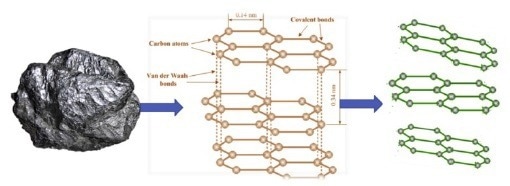
Using Spectroscopic Reflectometry to Characterize Graphene

Covalent Diamond–Graphite Bonding: Mechanism of Catalytic Transformation
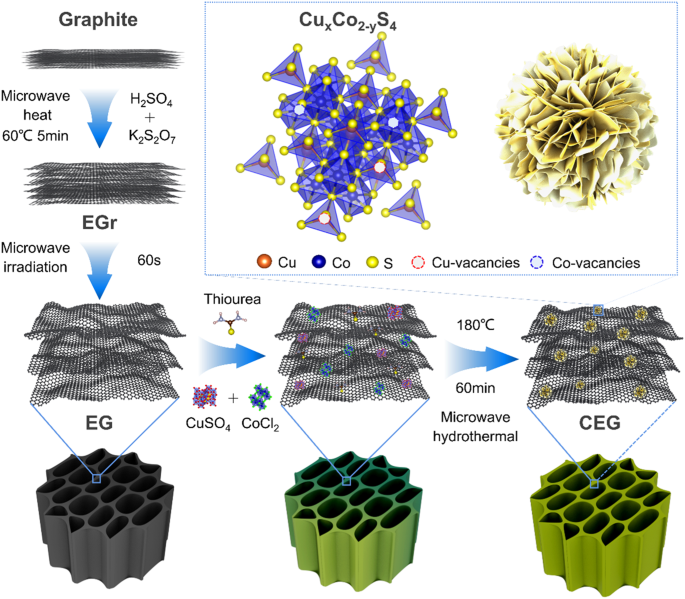
Synthesis of CuCo2S4@Expanded Graphite with crystal/amorphous heterointerface and defects for electromagnetic wave absorption

How to improve the natural graphite anode performance-Tycorun Batteries

Graphite - Wikipedia

Graphite - Definition, Structures, Applications, Properties, Use with Videos and FAQs of Graphite









