
BF can be obtained by reaction of $\mathrm{BF}_3$ with boron
4.7
(442)
Write Review
More
$ 19.00
In stock
Description
Why does boron trifluoride (BF3) act as a Lewis base and ammonia (NH3) acts as a Lewis acid? - Quora

Syntheses of Tricyanofluoroborates M[BF(CN)3] (M = Na, K): (CH3)3SiCl Catalysis, Countercation Effect, and Reaction Intermediates

⏩SOLVED:Boron trifluoride, BF3, and diethyl ether, (C2 H5)2 O, react…

Describe the shapes of BF3 and BH−4. Assign the hybridisation of boron in these species.
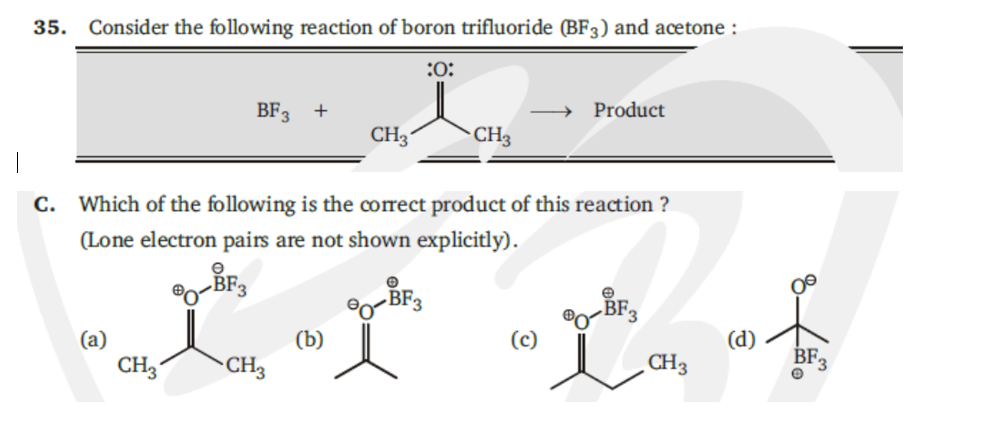
Solved 35. Consider the following reaction of boron

BF3-Activated Oxidation of Alkanes by MnO4

Setup for investigation of plasma chemical synthesis of B4C from

Boron Trifluoride - an overview


Boron Fluoride - an overview

a Reactions of formation of bulk boron carbide; b scheme of deposition


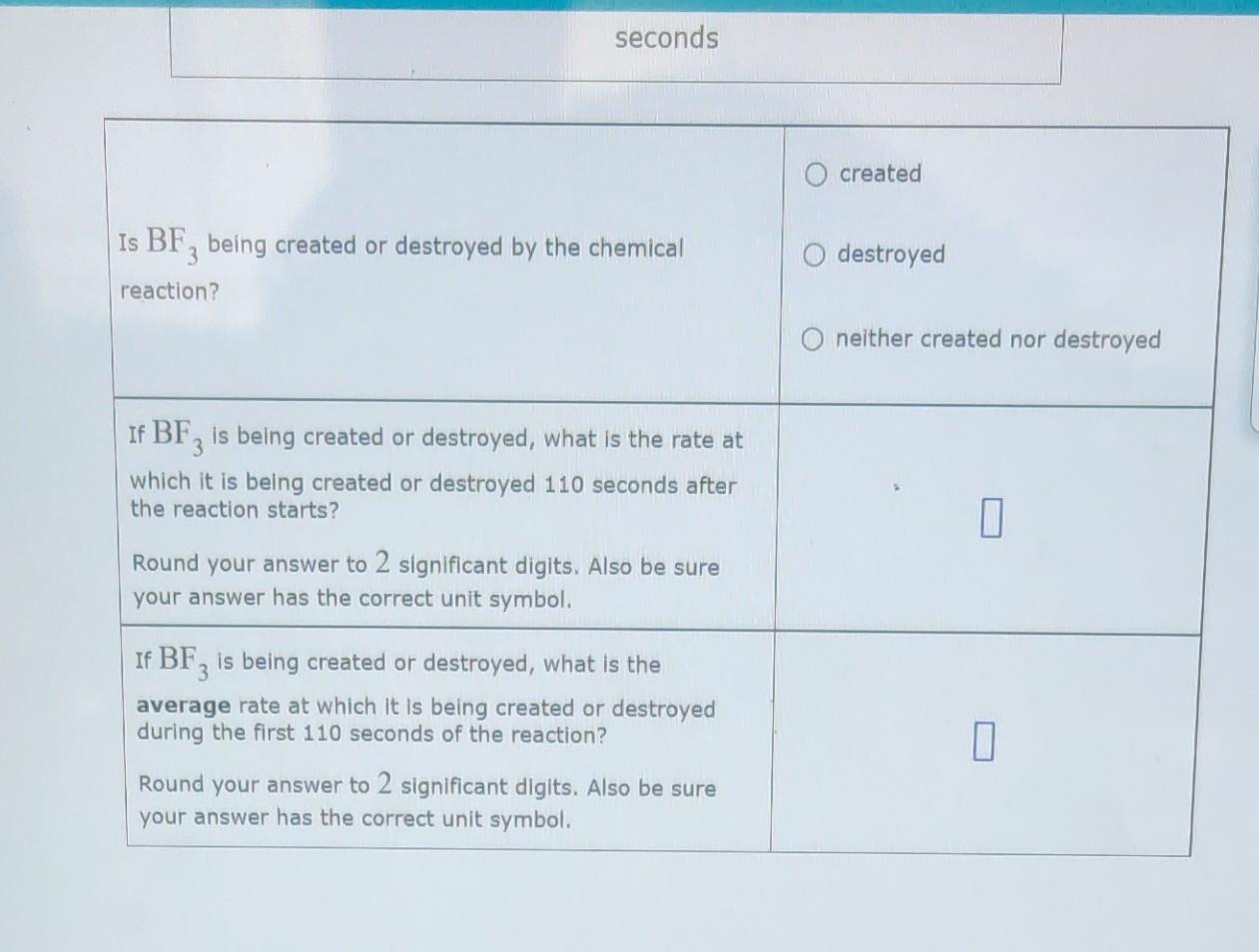
Solved Here is a graph of the pressure of boron trifluoride
Related products
You may also like









